Men B: its introduction into the routine schedule
Key learning points:
– Overview of the meningococcal bacteria
-Understand the development of the Men B vaccine
– To have a confident discussion with parents about the use of paracetamol with the MenB vaccine
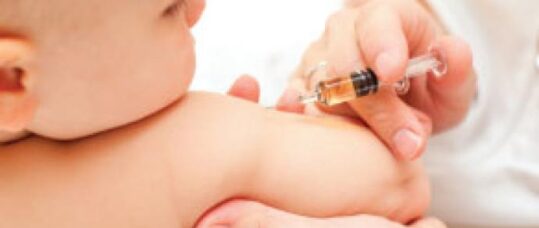
In March 2014 the UK Joint Committee for Vaccination and Immunisation (JCVI) published a position statement recommending the inclusion of the new meningococcal B (Men B) vaccine, Bexsero (also referred to as 4CMenB) in the routine infant immunisation programme.1 The vaccine has been part of the routine schedule since 1 September 2015. Primary care nurses and health visitors will play a vital role in promoting the vaccine to parents.
This article provides background information to allow practice nurses and health visitors to have a confident discussion with parents and address any questions or concerns they may have.
Meningococcus
Invasive meningococcal disease (IMD) occurs after infection with the bacterium Neisseria meningitidis, also known as meningococcus. Meningococci are classified according to the polysaccharide (sugary) capsule on the outside of the bacteria and by the proteins found on the outer membrane.2,3
Twelve serogroups have been identified, of which capsular groups A, B, C, W and Y are the most common. IMD caused by capsular group C has almost been eradicated from countries with effective immunisation schedules, such as England and Wales.4 For the year 2013/2014 there were 636 confirmed cases of IMD, an incidence of about one per 100,000 in England, with a case fatality of 6%.5 Capsular group B accounts for nearly 70% of IMD and the majority of cases in infants (85%) and toddlers (92%). The ages of highest incidence are infancy and one to four year olds, with a further peak at 15 to 19 years of age.3,5,6
During the last two years there has been an increase in cases caused by capsular group W. The JCVI, in response to this, advised an urgent catch up vaccination programme to vaccinate all adolescents and first time university entrants up to 25 years with a Men A,C,W,Y vaccine; this commenced 1 August 2015.7
Meningitis and septicaemia
IMD includes meningitis or septicaemia or more usually a combination of both. The bacteria enters the blood stream by penetrating the mucosa of the nose and throat. In the bloodstream the bacteria multiply rapidly. In meningitis, the bacteria crosses the blood-brain barrier causing inflammation of the meninges, whereas septicaemia is the blood poisoning form of the disease.8
Around one-in-eight survivors have long-term complications, including brain damage, deafness, epilepsy, limb/digit loss or cognitive deficit.3 Meningitis and septicaemia caused by the meningococcal bacteria can be treated with antibiotics. However, because the bacteria multiplies so rapidly it can be just hours between the first signs of the disease and someone becoming critically unwell.9 Making an early diagnosis is essential for successful treatment. Immunisation is the only rational approach to prevent disease burden and serious outcomes of infection.4,10
Meningococcal carriage
Although IMD continues to have devastating outcomes it is relatively rare. It is common for most individuals infected by the bacteria to have a period of asymptomatic carriage, where the bacteria colonise the nasopharynx and then the body’s immune system clear it again.11 Carriage rates vary according to age, from 4.5% in childhood to a peak of 23.7% in 19 year olds, before decreasing again in adulthood.11
Related Article: Postnatal contraception advice reduces the risk of back-to-back pregnancies
The recent introduction of the Men A,C,W,Y vaccine programme is an example of a strategy with the potential to not only protect vaccinated individuals but also to build ‘herd protection’. Vaccinating adolescents prior to them acquiring the bacteria for carriage, could potentially impact carriage in this high-risk age group that currently acts as a reservoir of meningococcal disease.12
If immunisation with the 4CMenB disrupts the carriage of the bacteria, this could reduce transmission to unimmunised populations, generating herd protection.11,12 This is, at present, an area of ongoing research.13
4CMenB vaccine
Making a Men B vaccine that is immunogenic (able to trigger a production of antibodies) has taken a long time and has used novel approaches. The outer polysaccharide capsule of the capsular group B meningococcus (Men B), which is used for the development of other vaccines including the Men C vaccine, is structurally identical to foetal brain cell molecules, making the meningococcal B capsule poorly immunogenic as the body does not recognise it as being foreign.14
Immune response
In clinical trials 5,849 infants and children under two years of age, 250 children aged between two and 10 years of age and 2,677 adolescents and adults have received at least one dose of the 4CMenB vaccine.15 The results show that 4CMenB is immunogenic in all ages, producing high concentrations of antibodies that can kill most meningococcal B strains.13,15,16,17,18
We know the vaccine produces an individual protective response against the strains that were used to make the vaccine. However, what is unclear is how effective it will be when introduced in the routine schedule against the broad number of strains circulating in the UK and the impact it will have on herd immunity and carriage. Phase 3 clinical trials assessing the overall effectiveness of a vaccine would have provided further evidence to support the introduction of the vaccine. However, as the disease is so rare studies would need to randomise hundreds of thousands of participants, making this impossible in practice.19
A system developed to predict overall strain coverage, MATS (Meningococcal Antigen Typing System), suggests that up to 88% of strains identified in England and Wales would be killed by the Men B vaccine.20
Prophylactic antipyretics
The outer membrane vesicle (OMV) component of the Men B vaccine enhances the immune response, this results in a reactogenic vaccine.4 To counteract the reactions after vaccination the JCVI recommended that three doses of paracetamol should be given in the 24 hours following the two-month and four-month primary immunisations (when 4CMenB is given at the same time as the other routine infant immunisations).1
The impact of prophylactic paracetamol on the occurrence of fever, other local reactions and immune responses was examined. The results showed that oral paracetamol given at the time of vaccination, then two subsequent doses at four to six hour intervals, significantly reduced the incidence of febrile reactions ≥38.5°C and fewer infants experienced local reactions without compromising immune responses.
This information can be used by practice nurses and health visitors to reassure parents in the administration of the 4CMenB vaccine. Guidance on the use of paracetamol and communicating with parents can be found in Immunisation against infectious disease(commonly called ‘The Green Book’)2 and on the Public Health England (PHE) website.21 A useful video of a mock consultation with a parent; explaining the changes in advice around prophylactic paracetamol use was developed by the Oxford Vaccine Group and Public Health England.22
JCVI recommendation
The indication for 4CMenB in the summary of product characteristics (SPC)15 is for three doses for infants from two months of age with a booster at 12 months.17
The JCVI stated that two doses of vaccine given two months apart is likely to provide a similar antibody response as that achieved by three doses one month apart, providing protection before the peak incidence of disease at five months of age.1
Implications for practice
The vaccine has been introduced into the schedule at two, four and 12 months for infants born on or after 1 July 2015. See the Resources section for details regarding the catch up programme, managing children who present late or transfer from another country, and for individuals with specific medical conditions.
Conclusion
The new Men B vaccine is immunogenic; it has a good safety profile, although due to the OMV component it is reactogenic, causing fever when given with other routine immunisations. Three doses of prophylactic paracetamol significantly reduces fever, without an effect on the immune response.
There is a need to educate parents to prepare them for the possibility of a fever after vaccination, so that the health service is not impacted by a rise in the number of attendances at GP surgeries or A&E departments. However, babies with an unexplained fever should still have assessment by a physician. Clear communication and instructions in fever management will alleviate concern for parents, thus avoiding low uptake of subsequent vaccinations.
The routine Men B vaccine can save lives and practice nurses and health visitors play a vital role in ensuring all infants benefit from this intervention through their informed discussions with parents.
Resources
Immunisation against infectious disease book –
gov.uk/government/uploads/system/uploads/attachment_data/file/462629/2904512_Green_Book_Chapter_22_v6_0W.PDF
References
Related Article: ‘Concerning acceleration’ in drug-resistant gonorrhoea ahead of vaccine programme
1. Department of Health and Public Health England. Joint Committee on Vaccination and Immunisation position statement on use of Bexsero® meningococcal B vaccine in the UK, 2014. gov.uk/government/publications/meningococcal-b-vaccine-jcvi-position-statement (accessed 23 September 2015).
2. Public Health England. Immunisation against infectious disease, 2014. gov.uk/government/uploads/system/uploads/attachment_data/file/462629/2904512_Green_Book_Chapter_22_v6_0W.PDF (accessed 23 September 2015).
3. Public Health England.Introducing two new national meningococcal immunisation programmes in the UK: i) Infant MenB Programme ii)Teenage MenACWY programme. gov.uk/government/publications/menb-and-menacwy-programmes-advanced-training-slides (accessed 23 September 2015).
4. Andrews S, Pollard A. A Vaccine against serogroup B Neisseria meningitides: dealing with uncertainty. Lancet Infectious Disease 2014;14:426-34 (accessed 29 September 2015).
5. Public Health England. Infection report Invasive meningococcal disease: 2013/2014 annual data by epidemiological year, 2015. gov.uk/government/uploads/system/uploads/attachment_data/file/397913/hpr0315_imd.pdf (accessed 29 September 2015).
6. Public Health England.Invasive Meningococcal Disease data slide set: 2005 and 2006 to 2014 and 2015 epidemiological years, 2015.
gov.uk/government/publications/meningococcal-disease-laboratory-confirmed-cases-in-england-and-wales (accessed 23 September 2015).
7. Public Health England. Introduction of a meningococcal ACWY immunisation programme for adolescents: Information for healthcare professionals. gov.uk/government/uploads/system/uploads/
attachment_data/file/451525/PHE_MenACWY_Immunisation_
programme_August_2015.pdf (accessed 23 September 2015).
8. Knight C, Glennie L. Early recognition of meningitis and septicaemia Journal of family health, 2010. https://pubmed.ncbi.nlm.nih.gov/20397549/
9. Meningitis now. Signs and Symptoms. meningitisnow.org/meningitis-explained/signs-and-symptoms/ (accessed 23 September 2015).
10. Pace D, Pollard A. Meningococcal disease: Clinical presentation and sequelae. Vaccine, 2011. elsevier.com/locate/vaccine (accessed 1 October 2015).
11. Christensen H, May M, Bowen L, Hickman M, Trotter CL.
Meningococcal carriage by age: a systematic review and meta-analysis. Lancet Infectious diseases 2010;10:853-61. DOI:10.1016/S1473-3099(10)70251-6.
12. Nolan T, O’Ryan M, Wassilc J, Abitbold V, Dull P. Vaccination with a multicomponent meningococcal B vaccine in prevention of disease in adolescents and young adults. Vaccine 2015;33:4437-4445. ncbi.nlm.nih.gov/pubmed/26187261 (accessed 23 September 2015).
13. McQuaid F, Snape M, John T, Kelly S, Robinson H, Yu L, Toneatto D, D’Agostino D, Dull P, Pollard A. Persistence of specific bactericidal antibodies at 5years of age after vaccination against serogroup B meningococcus in infancy and at 40 months. Canadian Medical Association Journal2015; 21;187(7):E215-23. DOI:10.1503 /cmaj.141200 (accessed 23 October 2015).
14. Tan L, Carlone G, Borrow R. Advances in the development of vaccines against Neisseria meningitides. The New England Journal of Medicine 2010;362:1511-20. DOI:10.1056/NEJMra0906357 (accessed 23 October 2015).
15. Electronic Medicines Compendium (EMC). Summary of Product Characteristics. Bexsero Meningococcal Group B vaccine for injection. medicines.org.uk/emc/medicine/28407 (accessed 23 September 2015).
Related Article: Action needed to tackle untreated hearing loss in care homes
16. Gossger N, Snape M, Yu LM, Finn A, Bona G, Esposito S, Principi N, JDiez-Domingo J, Sokal E, Becker B, Kieninger D, Prymula R, Dull P, Ypma E, Toneatto D, Kimura A, Pollard A. Immunogenicity and tolerability of recombinant serogroup B meningococcal vaccine administered with or without routine infant vaccinations according to different immunization schedules: a randomized controlled trial. JAMA 2012;307(6):573-82.
17. Santolaya M, O’Ryan M, Valenzuela M, Prado V, Vergara R, Muñoz A, Toneatto,D, Graña G, Wang H, Clemens R, Dull P. Immunogenicity and tolerability of a multicomponent meningococcal serogroup B (4CMenB) vaccine in healthy adolescents in Chile: a phase 2b/3 randomised, observer-blind, placebo-controlled study. Lancet2012;379(9816):617-24. DOI:10.1016/ S0140-6736(11)61713-3 (accessed 23 October 2015).
18. Public Health England correspondence. Introduction of MenB vaccination for infants. gov.uk/government/publications/menb-
vaccination-introduction-from-1-september-2015 (accessed 23 September 2015).
19. Ed. The case for vaccinating against meningitis B. The Lancet Infectious Diseases2014;14(5):359. DOI: 10.1016/S1473-3099(14)70756-X (accessed 23 October 2015).
20. Frosi G, Biolch A, Sapio M, Rigat F, Gilchrist S, Lucidarme J, Findlow J, Borrow R, Pizza M, Giuliani M, Medini D. Bactericidal antibody against a representative epidemiological meningococcal serogroup B panel confirms that MATS underestimates 4CMenB vaccine strain coverage. Vaccine 2013;31:4968 4974. sciencedirect.com/science/article/pii/S0264410X13010906 (accessed 1 October 2015).
21. Public Health Guidance. MenB vaccine and paracetamol, 2015. gov.uk/government/publications/menb-vaccine-and-paracetamol. (accessed on 12 October 2015).
22. Oxford Vaccine Group. Talking to parents about MenB vaccine: a film for healthcare professionals. ovg.ox.ac.uk/menb-training-film (accessed 14 October 2015).

See how our symptom tool can help you make better sense of patient presentations
Click here to search a symptom


In March 2014 the UK Joint Committee for Vaccination and Immunisation (JCVI) published a position statement recommending the inclusion of the new meningococcal B (Men B) vaccine, Bexsero (also referred to as 4CMenB) in the routine infant immunisation programme



