Top tips on prescribing hormone replacement therapy

1. Know the basics and be able to explain them to patients
Symptoms of the menopause occur due to oestrogen deficiency after ovarian follicular activity ceases1; giving oestrogen alone would therefore treat these symptoms but leave the endometrium exposed to unopposed oestrogen. Over time, this causes endometrial proliferation as cells multiply in response to the oestrogen, and increases the risk of endometrial cancer. Women who have their uterus therefore need combined hormone replacement therapy (HRT) with oestrogen and a progestogen, whereas those who have had a hysterectomy can have oestrogen alone. Seek advice in the case of women who have had a hysterectomy due to endometriosis, as they may also need combined HRT for a period after surgery. Women who have only vaginal symptoms (such as atrophy/dryness) may benefit from vaginal oestrogen alone; in this situation there is no need to take a progestogen even for women with a uterus. Vaginal oestrogen can also be added to systemic HRT if genital symptoms remain a problem.
Key symptoms of the menopause:
- Change to menstrual pattern
- Hot flushes/night sweats
- Cognitive impairment and mood disorders
- Urogenital symptoms
- Altered sexual function
- Sleep disturbance
- Other: joint and muscle pain, headache, fatigue
2. Be able to talk about the risks in simple terms
Levels of health literacy in the general public are low – more than 50% of working-age adults can’t understand or make use of everyday health information if it involves the use of numeracy skills.2 Offering information in easy-to-use chunks and using infographics can help your patient to make an informed decision – the table ‘Explaining the risks’ below may help. There are few absolute contraindications to HRT but it should not be initiated in primary care to women with a history of breast cancer – a careful and documented discussion should be had with the oncologist before the prescription of any HRT, systemic or vaginal. These women may benefit from a non-hormonal alternative such as clonidine, gabapentin or venlafaxine.1,3
Explaining the risks
Breast cancer4
- HRT is not associated with an increased risk of dying from breast cancer.
- NICE guidance is based on studies indicating there would be four extra cases of breast cancer for every 1,000 women taking combined HRT for five years, and four fewer cases for every 1,000 women taking oestrogen-only HRT for five years.
- A 2019 meta-analysis5 raised these figures to 20 cases per 1,000 for combined HRT with daily progestogen, 14 per 1,000 for combined HRT with intermittent progestogen and five per 1,000 for oestrogen-only HRT.
- That study was heavily criticised because of flaws in its method and because most of the women used old HRT regimes; the ones currently in use are associated with a lower risk.
- Women may find the British Menopausal Society’s infographic helpful. It compares the risk of HRT with other factors such as weight, alcohol intake and exercise.
Blood clots
Related Article: Postnatal contraception advice reduces the risk of back-to-back pregnancies
- There is no increased risk of venous thromboembolism (VTE) if HRT is given transdermally (via patch or gel).1
- The risk of VTE is approximately doubled over the baseline if oral HRT is used4; transdermal HRT is increasingly used as standard and should certainly be the first choice for women with a BMI over 30.
Cardiovascular disease and diabetes
- If started under the age of 60, the use of HRT does not increase the risk of cardiovascular disease or diabetes, and the presence of risk factors such as smoking, high cholesterol or pre-existing diabetes is not a contraindication to HRT use.4
Ovarian cancer1,4
- There is a very small increase – around one additional case per 1,000 women and one additional death per 1,700 women who use HRT for five years.
3. Balance the risks against the benefits
The immediate benefit to women is the relief of symptoms. If symptoms are so bad that it is affecting a woman’s job, home life, ability to exercise and sleep, then it is likely that she will benefit from HRT purely on the basis of symptom control. HRT use also has longer-term benefits in terms of a reduction in the risk of fragility fractures1, increase in muscle mass1 and strength, and a reduction in all-cause mortality.4
4. Don’t forget contraception
A woman will need contraception for two years after her last period if it occurs before the age of 50 and for one year if it occurs after 50.1 HRT is not contraceptive but the Mirena intrauterine system (IUS) can be used for both contraception and the progestogenic component of HRT, making it an excellent contraceptive choice for women in their 40s.6
5. Be clear about cyclical versus continuous HRT
It is perfectly acceptable to prescribe during the perimenopause. This is defined by NICE1 as: ‘The period before the menopause when the endocrinological, biological, and clinical features of approaching menopause start. It is characterised by irregular cycles of ovulation and menstruation and ends 12 months after the last menstrual period.’
Women who use HRT within one year of their last menstrual period (LMP) should generally have cyclical or sequential HRT, where the use of a progestogen for two weeks of the month gives a regular bleed. Moving straight to continuous HRT during the perimenopause risks irregular bleeding. If starting more than one year after the last period, then continuous HRT can be used. The exception to this is women who use the Mirena IUS – this can be given as the progestogenic component of HRT at any stage. Mirena is the only brand of IUS that is licensed for HRT – no other brand should be used. The HRT licence for Mirena is for four years, one year less than the contraceptive licence, but the guidance is that it can be used off-licence for this indication for 5 years.6
Related Article: ‘Concerning acceleration’ in drug-resistant gonorrhoea ahead of vaccine programme
There is a bewildering variety of HRT preparations on the market, although there have been some well-documented shortages over the past few years.7 If you follow the basic principles discussed above and outlined in the flowchart below, then your choice may be guided as much by what your local pharmacy can get as by anything else.
My regimen of choice is oestradiol gel with a Mirena IUS or micronised progestogen for women who have a uterus. If the oestrogen and progestogen are given separately in this way then it is vital the woman understands why the progestogen is needed – otherwise there is a risk she will realise the oestrogen alone controls her symptoms, stop using the progestogen and inadvertently increase her risk of endometrial cancer.
Further information on the details of prescribing is available in the Primary Care Women’s Health Forum guide to HRT4, on the NICE CKS page1 and from MIMS, which has a useful table of HRT preparations.8
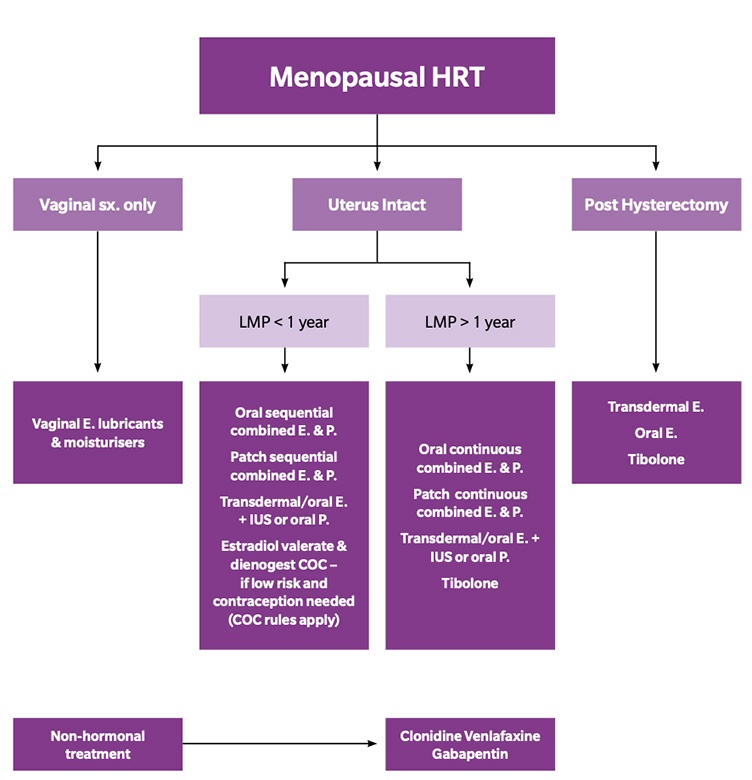
Key: E=Oestrogen; P=Progestogen; IUS=intrauterine system; LMP=last menstrual period
Declaration of interest: Dr Hazell is on the executive committee of the PCWHF, a role that involves both paid and paid work
Related Article: Action needed to tackle untreated hearing loss in care homes
References
(All links accessed on 16 June, 2021)
- NICE CKS: Menopause. London: NICE 2020.
- Public Health England. Improving health literacy to reduce health inequalities. London: PHE, 2015.
- British Menopause Society. Prescribable alternatives to HRT. Buckinghamshire: BMS, 2018.
- Primary Care Women’s Health Forum. Menopause – guidance on management and prescribing HRT. Bedfordshire: PCWHF, 2020.
- Collaborative Group on Hormonal Factors in Breast Cancer. Type and timing of menopausal hormone therapy and breast cancer risk. Lancet 2019;394(1024):1159-1168.
- Faculty of Sexual Health and Reproductive Healthcare. Clinical guideline: Contraception for women aged over 40 years. London: FSRH, 2019.
- Cox T. Pharmacies suffered shortages in every major HRT brand pre-Covid-19. Chemist and Druggist, May 2020.
- Hormone replacement therapy table: doses and costs of HRT preparations. MIMS, May, 2021.

See how our symptom tool can help you make better sense of patient presentations
Click here to search a symptom


There is a bewildering variety of HRT preparations on the market, although there have been some well-documented shortages over the past few years.



